Carrow, R.N., and R. Duncan. 2011. Best management practices for saline and sodic turfgrass soils: Assessment and reclamation. p. 79. CRC Press, Boca Raton, Fla.
Carrow, R.N., D.V. Waddington and P.E. Rieke. 2001a. Turfgrass soil fertility and chemical problems. p. 251. Wiley, Hoboken, N.J.
Carrow, R.N., D.V. Waddington and P.E. Rieke. 2001b. Turfgrass soil fertility and chemical problems. pp. 354-355. Wiley, Hoboken, N.J.
Carrow, R.N., D.V. Waddington and P.E. Rieke. 2001c. Turfgrass soil fertility and chemical problems. p. 253. Wiley, Hoboken, N.J.
Carrow, R.N., D.V. Waddington and P.E. Rieke. 2001d. Turfgrass soil fertility and chemical problems. p. 251. Wiley, Hoboken, N.J.
Carrow, R.N., D.V. Waddington and P.E. Rieke. 2001e. Turfgrass soil fertility and chemical problems. p. 242. Wiley, Hoboken, N.J.
Christians, N.E. 2011. Fundamentals of turfgrass management. p. 163. Wiley, Hoboken, N.J.
Fagerness, M., S. Keeley and D. Whitney. 1998. A guide to turfgrass nutrient recommendations. Kansas State University Cooperative Extension bulletin MF-2311.
National Atmospheric Deposition Program. 2016. http://nadp.sws.uiuc.edu/data/NTN/
Rosen, C.J., P.M. Bierman, and R.D. Eliason. 2008. University of Minnesota Extension. https://conservancy.umn.edu/handle/11299/49265
Soltanpour, P.N., and R.H. Follett. 2007. Soil test explanation. Colorado State University Cooperative Extension Service. http://extension.colostate.edu/topic-areas/ agriculture/soil-test-explanation-0-502/
Published with support from the NDSU Extension Master Gardener program.
 The NDSU Extension Service does not endorse commercial products or companies even though reference may be made to tradenames, trademarks or service names. NDSU encourages you to use and share this content, but please do so under the conditions of our Creative Commons license. You may copy, distribute, transmit and adapt this work as long as you give full attribution, don’t use the work for commercial purposes and share your resulting work similarly. For more information, visit www.ag.ndsu.edu/agcomm/creative-commons.
The NDSU Extension Service does not endorse commercial products or companies even though reference may be made to tradenames, trademarks or service names. NDSU encourages you to use and share this content, but please do so under the conditions of our Creative Commons license. You may copy, distribute, transmit and adapt this work as long as you give full attribution, don’t use the work for commercial purposes and share your resulting work similarly. For more information, visit www.ag.ndsu.edu/agcomm/creative-commons.
For more information on this and other topics, see www.ag.ndsu.edu
County commissions, North Dakota State University and U.S. Department of Agriculture cooperating. NDSU does not discriminate in its programs and activities on the basis of age, color, gender expression/identity, genetic information, marital status, national origin, participation in lawful off-campus activity, physical or mental disability, pregnancy, public assistance status, race, religion, sex, sexual orientation, spousal relationship to current employee, or veteran status, as applicable. Direct inquiries to Vice Provost for Title IX/ADA Coordinator, Old Main 201, NDSU Main Campus, 701-231-7708, ndsu.eoaa@ndsu.edu. This publication will be made available in alternative formats for people with disabilities upon request, 701-231-7881. web-4-23


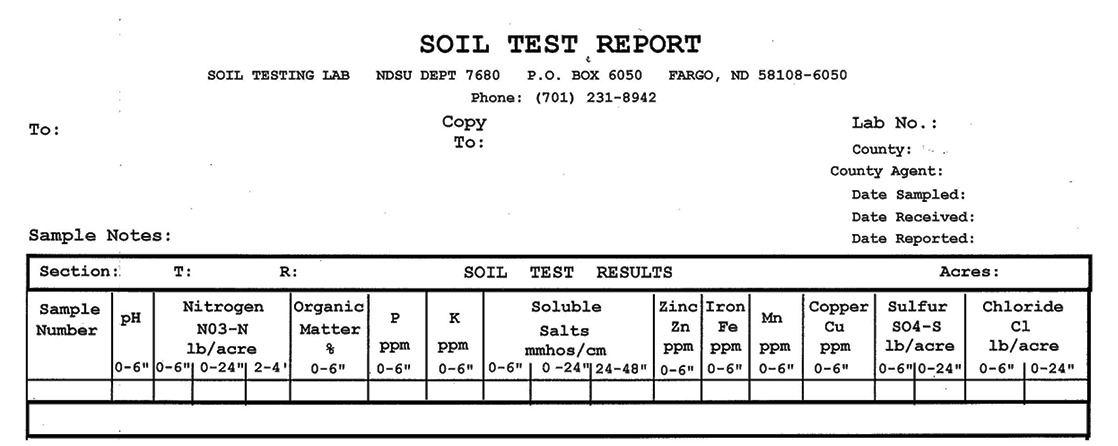

 The NDSU Extension Service does not endorse commercial products or companies even though reference may be made to tradenames, trademarks or service names. NDSU encourages you to use and share this content, but please do so under the conditions of our Creative Commons license. You may copy, distribute, transmit and adapt this work as long as you give full attribution, don’t use the work for commercial purposes and share your resulting work similarly. For more information, visit
The NDSU Extension Service does not endorse commercial products or companies even though reference may be made to tradenames, trademarks or service names. NDSU encourages you to use and share this content, but please do so under the conditions of our Creative Commons license. You may copy, distribute, transmit and adapt this work as long as you give full attribution, don’t use the work for commercial purposes and share your resulting work similarly. For more information, visit 